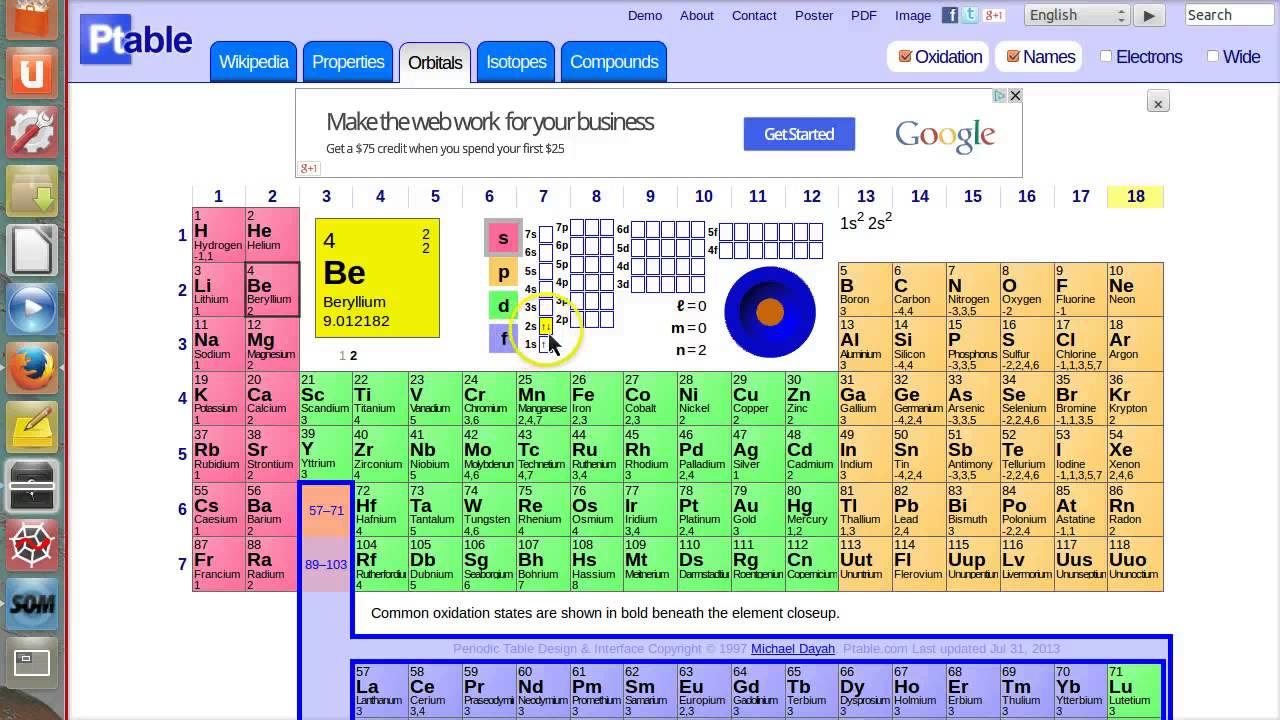
Other important groupings of elements in the periodic table are the main group elements, the transition metals, the lanthanides, and the actinides. The periodic table is divided into groups (. Metals are lustrous, good conductors of electricity, and readily shaped (they are ductile and malleable), whereas solid nonmetals are generally brittle and poor electrical conductors. To find the number of valence electrons for an element, you need to look at its position in the periodic table. They are separated by a diagonal band of semimetals. Metals are located on the left of the periodic table, and nonmetals are located on the upper right. Semimetals exhibit properties intermediate between those of metals and nonmetals. 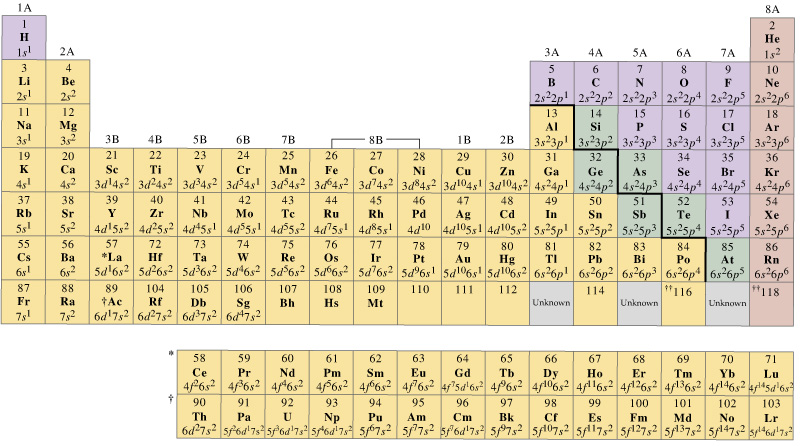
The elements can be broadly divided into metals, nonmetals, and semimetals. Some of the groups have widely-used common names, including the alkali metals (Group 1) and the alkaline earth metals (Group 2) on the far left, and the halogens (Group 17) and the noble gases (Group 18) on the far right. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods. Transition Metals: Groups 3-12 - d and f block metals have 2 valence electrons. Alkaline Earth Metals: Group 2 (IIA) - 2 valence electrons. With the exception of the transition metals, the units digit of the group number identifies how many valence electrons are associated with a neutral atom of an. It arranges of the elements in order of increasing atomic number. Another common method of categorization recognizes nine element families: Alkali Metals: Group 1 (IA) - 1 valence electron. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. The periodic table is used as a predictive tool. Valence electrons, the electrons in the outermost or valence shell, are important as they provide insight into an elements chemical properties and are the ones. As expected, semimetals exhibit properties intermediate between metals and nonmetals. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Valence electrons: The electrons present in the outermost orbit of an atom are called valence electrons. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. We can write the configuration of oxygen's valence electrons as 2s2p. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. Transcript Valence electrons are the electrons in the outermost shell, or energy level, of an atom. 
The number of valence electrons in an atom is reflected by its position in the periodic table of the elements (see the periodic table in the Figure below). 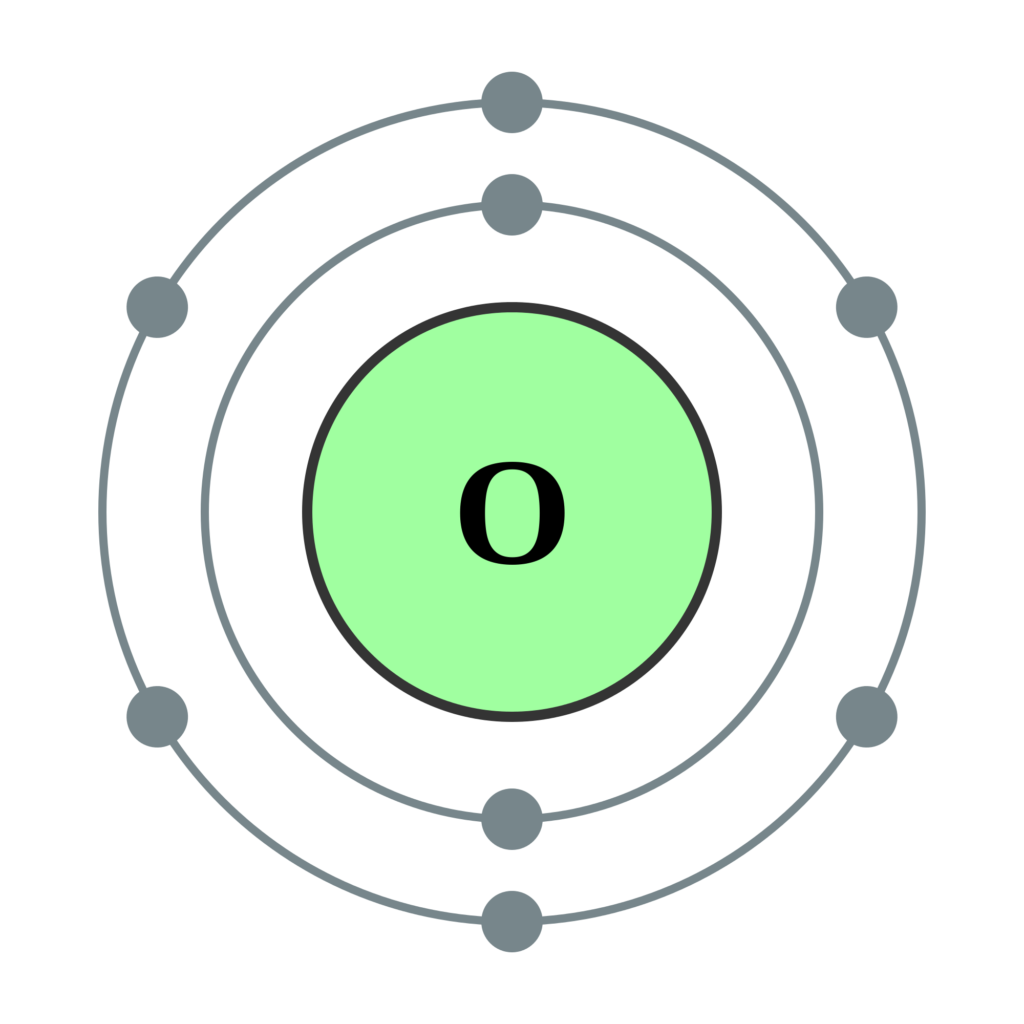
The vast majority of the known elements are metals. Valence Electrons and the Periodic Table. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. But the valency of elements, when combined with H or O first, increases from 1 to 4 and then it reduces to zero. While moving left to right across a period, the number of valence electrons of elements increases and varies between 1 to 8. Variation Of Oxidation State Along a Period. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Periodic Trends in the Oxidation States of Elements. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. For elements after No, the electron configurations are tentative.\) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line). The electron configurations of the elements indicated in blue are also anomalous, but the reasons for the observed configurations are more complex. The electron configurations of elements indicated in red are exceptions due to the added stability associated with half-filled and filled subshells. \): Electron Configurations of the Elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
